 We address this shortcoming with a mathematical model representing the organelle as a cell-wide dynamical network subjected to opposing actions of fission and internal fusion – processes known experimentally but not yet accurately specified. Despite extensive knowledge of mitochondrial physiology, accurate description of their large-scale architecture is missing, partly due to substantial variability of reticulum geometries found in different cell types, and a capability for fast radical changes. Most often, they form a constantly reshaping tubular reticulum spread over the cytosol. Mitochondria control energy production, initiation of cell death and several other critical cellular processes. Analysis of the reticulum adaptive configuration offers a direct clarification for its impact on numerous physiological processes strongly dependent on mitochondrial dynamics and organization, such as efficiency of cellular metabolism, tissue differentiation and aging. It forms a basis for understanding the architecture of mitochondria as a cell-wide but inhomogeneous organelle. The detailed size distribution of the network components predicted by the dynamic graph representation introduces a relationship between chondriome characteristics and cell function. Intrinsic morphological instability of the mitochondrial reticulum resulting from its vicinity to the percolation transition is proposed as a novel mechanism that can be utilized by cells for optimizing their functional competence via dynamic remodeling of the chondriome. The computational analysis indicates that mitochondrial networks exhibit a percolation threshold. Using both mean-field deterministic and explicit stochastic mathematical methods we establish a relationship between the chondriome structural network characteristics and underlying kinetic rate parameters. Subsequently, we introduce a graph-based model of the chondriome able to encompass its inherent variability in a single framework. On the basis of experimentally determined structural features we establish the tip-to-tip and tip-to-side fission and fusion events as dominant reactions in the motility of this organelle. Here we address the principles of formation and the large-scale organization of the cell-wide network of mitochondria. Currently, quantitative understanding of its morphological characteristics is largely absent, despite major progress in deciphering the molecular fission and fusion machineries shaping its structure. 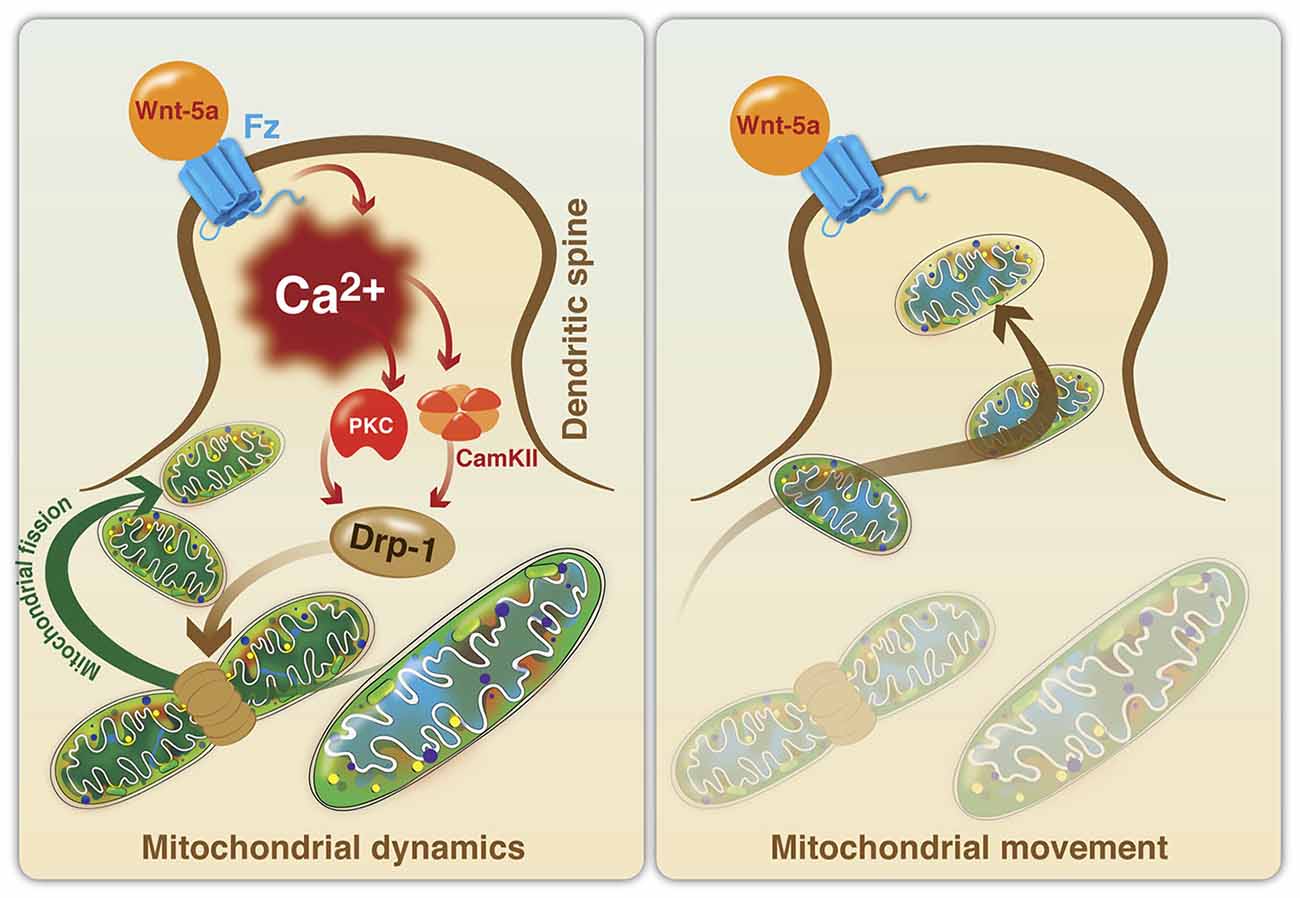
© 2021 Federation of American Societies for Experimental Biology.Mitochondria form a dynamic tubular reticulum within eukaryotic cells. This review discusses the molecular mechanisms that control mitochondrial fusion and fission and how the disruption of mitochondrial dynamics manifests into various disease conditions.ĭiseases dynamics fission fusion mitochondria. The imbalance between these two processes is associated with various ailments such as cancer, neurodegenerative and cardiovascular diseases. 
On the other hand, fission is crucial for mitochondrial division and quality control. Mitochondrial fusion allows the transfer of gene products between mitochondria for optimal functioning, especially under metabolic and environmental stress. Thus, the maintenance of mitochondrial integrity and homeostasis is extremely critical, which is achieved through continual fusion and fission. Besides, they play an active role in calcium and damage-associated molecular patterns signaling, amino acid, and lipid metabolism, and apoptosis. Mitochondria are highly dynamic, maternally inherited cytoplasmic organelles, which fulfill cellular energy demand through the oxidative phosphorylation system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
